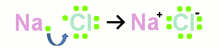By Kelsea Bergerson, Mackenzie Elmhirst, Luke Shroyer, and Marilynn Hunt
Overview
The objective of this project was to design an outdoor class space that would fit the needs of the majority of people on campus. Before we got to designing there was a lot of background work we needed to do, such as the client needs survey, site selection, and the materials testing lab. These things gave us more information on how to design our space. The entire project took us about six weeks to complete. This was a huge project that taught us about all the different aspects of engineering. We made schematics, models, a materials list, a budget, and a presentation. We also did a lot of mini labs like the hot water heater lab and the wind turbine lab. They helped teach us about clean, renewable energy sources. This was a long project, but it was worth the while.
Concepts
- Atoms, molecular structures, states of matter
- Specific heat capacity and ways heat is transferred (radiation, conduction, convection, insulation)
- Angles of the sun
- Laws of Thermodynamics
- Energy Justification
NOTE: pictures of binder paper notes are in the process of being replaced with typed notes.
1. Atoms, molecular structures, and states of matter
2. Ways heat is transferred and Specific heat capacity
|
Almost all energy for the Earth comes from the sun.
conduction: heat trasfer through solid. Some solids are better at transferring heat then others. (Ex. copper is better than aluminum) convection: transfer of heat through fluid (Ex. liquid or gas) radiation: transfer of heat through waves, rays, and particles. No medium required. insulation: opposite of conduction; bad at transferring heat. Wood is a better insulator, while table is a better conductor. Less dense=better insulator (more air bubbles) |
Specific Heat Capacity We used the equation Q=mc△T (heat energy= mass * specific heat * change in heat) to find the amount of thermal energy that is used to raise and/or lower the temperature of things. The specific heat capacity of objects vary. For example, water's specific heat is 4.179 Jules , while copper's is 0.385 Jules. |
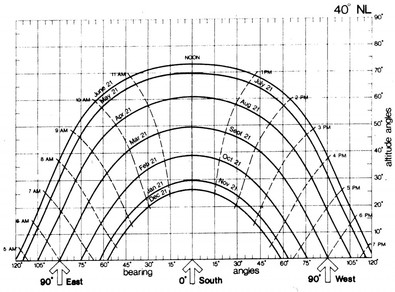
3. Angles of the sun
4. Laws of Thermodynamics
0 -explains temperature -if two systems are in thermal equilibrium with a third system, they are in equilibrium with each other (Ex. A=B and B=C, which means A=C)
1 -Conservation of energy -energy is neither created nor destroyed, but transferred from one form to another. Heat is a form of energy.
2 -Entropy increases -disorder increases our time, everything becomes the same temperature
3 -Temperature can never get to absolute zero. Heat always exists
1 -Conservation of energy -energy is neither created nor destroyed, but transferred from one form to another. Heat is a form of energy.
2 -Entropy increases -disorder increases our time, everything becomes the same temperature
3 -Temperature can never get to absolute zero. Heat always exists
5. Energy Justification
We should use less energy because fossil fuels are one of our biggest sources. The byproducts of them are bad for the environment. It gives off a lot of carbon dioxide and pollution. They are also nonrenewable energy sources, so we will eventually run out. Some good alternatives to fossil fuels are hydro power, solar power, and wind turbines. They are a few clean, renewable sources.
For more information, this is our energy justification spreadsheet.
For more information, this is our energy justification spreadsheet.
Reflection
This project was a lot of work. It was fun to get a life-like experience with engineering, but that also meant that we were constantly working on different things. I learned a lot about the way the world works and how many things you need to consider when designing any sort of structure or area. My favorite part of the project was designing the space and deciding what goes where.
Two things I think I did well was be committed and initiate progress on our project. I tried to stay on task and when we weren't finished on the due date, came in at lunch to work, and did some at home. I knew the Monday deadline and was adamant that we finished. I initiated by starting on parts of the project, suggesting to start things, and by asking people to do things. Towards the end of the project was when I did the most initiating because we were all getting tired.
I think the hardest part of the project for me was organization and time management. My binder was a mess. All of my papers were in different spots and I kept forgetting to number the pages. This made it hard to find certain pages. A way I can fix this problem is to number everything before I put it in my binder and to check the board that lists the order. Another thing I can do is always put new pages in the very back instead of the front. Time management was also an issue. My group and I would get something done, but after it was completed we wouldn't do anything for while. I think that since the project was so much longer than the others, we got tired of working on the same thing day after day. I was fine on days when we did labs because it spiced things up, but when we were working on our presentation I started losing interest. Some solutions to this could be doing different parts of the project each day instead of working on one thing a week straight. I should have varied what I worked on so I would do the schematics one day and the presentation the next.
Even though this was a very stressful project I did enjoy it. It took a lot of dedication and time. I had trouble with the materials list and pricing the cost, but it was a good thing to learn. I had no idea how many aspects there was to be an engineer. This project has definitely taught me the most out of all the projects we have done so far. I'm really excited to build the outdoor classroom soon!
Two things I think I did well was be committed and initiate progress on our project. I tried to stay on task and when we weren't finished on the due date, came in at lunch to work, and did some at home. I knew the Monday deadline and was adamant that we finished. I initiated by starting on parts of the project, suggesting to start things, and by asking people to do things. Towards the end of the project was when I did the most initiating because we were all getting tired.
I think the hardest part of the project for me was organization and time management. My binder was a mess. All of my papers were in different spots and I kept forgetting to number the pages. This made it hard to find certain pages. A way I can fix this problem is to number everything before I put it in my binder and to check the board that lists the order. Another thing I can do is always put new pages in the very back instead of the front. Time management was also an issue. My group and I would get something done, but after it was completed we wouldn't do anything for while. I think that since the project was so much longer than the others, we got tired of working on the same thing day after day. I was fine on days when we did labs because it spiced things up, but when we were working on our presentation I started losing interest. Some solutions to this could be doing different parts of the project each day instead of working on one thing a week straight. I should have varied what I worked on so I would do the schematics one day and the presentation the next.
Even though this was a very stressful project I did enjoy it. It took a lot of dedication and time. I had trouble with the materials list and pricing the cost, but it was a good thing to learn. I had no idea how many aspects there was to be an engineer. This project has definitely taught me the most out of all the projects we have done so far. I'm really excited to build the outdoor classroom soon!